This research was designed to compare the double closure system of the AMULET device with the WATCHMAN (an evidence-backed proven device).
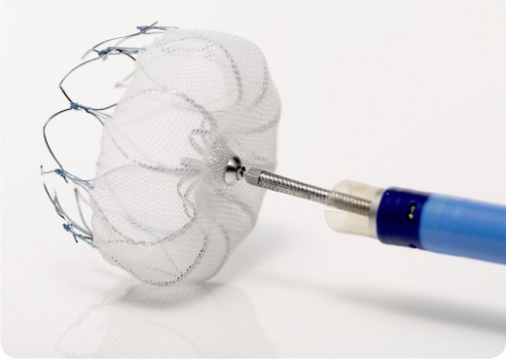
In that context, to prevent stroke in patients with non-valvular atrial fibrillation, the Amplatzer AMULET device was non-inferior in both safety and efficacy compared with the WATCHMAN. Atrial appendage occlusion in itself improved with AMULET, but with a higher rate of procedure-related complications that decreased with operator experience.
This study randomized 1878 patients with non-valvular atrial fibrillation and increased risk of stroke 1:1 to atrial appendage occlusion with either the AMULET or the WATCHMAN device.
There were two primary endpoints: one for safety (a composite of procedure-related complications, all-cause mortality, or major bleeding at 1 year) and one for efficacy (a composite of ischemic stroke or systemic embolism at 18 months), plus total appendage occlusion at 45 days.
The secondary endpoints were combinations of the aforementioned events.
The AMULET device was non-inferior to the WATCHMAN in the primary efficacy endpoint (14.5% vs. 14.7%; 95% confidence interval: 3.42 to 3.13; p < 0.001 for non-inferiority).
Major bleeding and all-cause mortality were also similar between devices (10.6% vs. 10.0% and 3.9% vs. 5.1%, respectively).
Read also: Asymptomatic Carotid Lesions and Cognitive Impairment: Does Intervention Play a Role?
The AMULET device almost doubled peri-procedural complications (4.5% vs. 2.5%)—primarily due to pericardial effusion and device embolization.
Major bleeding rates were similar in both groups (11.6% vs. 12.3%), while effective atrial appendage occlusion was higher with the AMULET device.
Conclusion
The AMULET atrial appendage occluder device was non-inferior in terms of safety and efficacy to the WATCHMAN device for stroke prevention in patients with non-valvular atrial fibrillation. Periprocedural complications observed with the AMULET decreased with operator experience.
Original Title: AMPLATZERTM AMULETTM LEFT ATRIAL APPENDAGE OCCLUDER VERSUS WATCHMANTM DEVICE FOR STROKE PROPHYLAXIS (AMULET IDE): A RANDOMIZED CONTROLLED TRIAL.
Reference: Dhanunjaya Lakkireddy et al. 10.1161/CIRCULATIONAHA.121.057063.
Subscribe to our weekly newsletter
Get the latest scientific articles on interventional cardiology





