The balloon-expandable valve has been granted approval in Europe for treatment of the complete risk spectrum of symptomatic severe aortic stenosis.
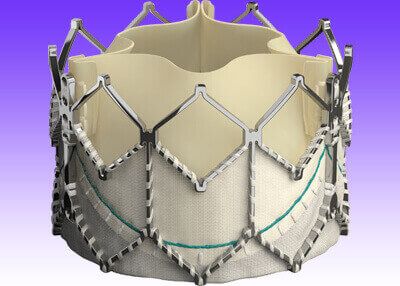
Europe has recently approved an expanded indication for the Sapien 3 system for use in low-risk patients, as reported by Edward Lifesciences.
Sapien 3, which includes the next-generation Sapien 3 Ultra, is the first transcatheter heart valve to receive CE Mark approval for patients with low surgical risk. The device is now approved for all patients, including those at high and intermediate risk, and those excluded from surgery.
The expanded indication for Sapien 3 is based on the results of the PARTNER 3 study, presented at the American College of Cardiology (ACC) 2019 Scientific Sessions last March and simultaneously published in the New England Journal of Medicine (NEJM).
That work included about 1000 low-risk patients, proving that transcatheter aortic valve replacement (TAVR) is associated with a significantly lower risk for the composite endpoint of death, stroke, and rehospitalization when compared with surgical valve replacement.
Last August, the US Food and Drug Administration (FDA), based on the same trial, approved Sapien 3 and Sapien 3 Ultra, and also included the self-expanding CoreValve, Evolut R, and Evolut PRO valves (all Medtronic), based on the results of the Evolut Low-Risk trial also presented at the ACC Scientific Sessions and published in NEJM.
So far, Medtronic is waiting for European approval, but that seems a mere administrative matter.
Once again, TAVR seems to travel backwards (in more ways than one) in relation to the path traveled by most devices used. It started with the most difficult patients and then reached the easiest subjects; it first obtained FDA approval and then CE Mark approval.
Original Title: Edwards Sapien 3 TAVI receives expanded approval in Europe.
Reference: Edwards Lifesciences. Published and accessed on: November 6, 2019.
Get the latest scientific articles on interventional cardiologySubscribe to our weekly newsletter
We are interested in your opinion. Please, leave your comments, thoughts, questions, etc., below. They will be most welcome.





