An FDA document recently published reminds doctors and patients of the need of annual follow up of abdominal aortic aneurysm patients receiving Endologix AFX endoprostheses, with imaging, and for life. The reason behind this recommendation is the risk of type III leaks.
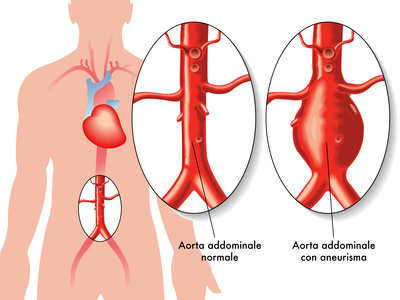
In 2019, the FDA had alerted us against this issue after finding a 2.5% cumulative chance of reintervention due to leaks type IIIa and IIIb.
More recently, the agency has recommended life-long followup and continues to evaluate data to determine whether the risk is even higher.
The latest data comes from a study published in The Journal of Vascular Surgery analyzing over 600 patients. After a 2-year followup, they saw 4% type 3 leaks with the AFX Strata, 5.1% with AFX Duraply and 14.1% with AFX2, even though it is difficult to assess this latest version of the device seeing the small number of patients involved.
The FDA also reminds us of two other presentations yet to be printed. In one of the studies, 25% of patients treated with AFX required reintervention, died or evolved with rupture aneurysms at 4-year followup. The other presentation showed markedly increased device rupture after 3 -years. With this information and the ongoing study, the FDA has been monitoring the postmarket of this device.
Read also: NT-proBNP Level Predicts Who May Benefit From TAVR.
The monitoring panel of endovascular devices are looking into type III leaks with the AFX, treatment options, complications, and future surveillance strategies for patients receiving this device to treat infrarenal abdominal aortic aneurysms.
UPDATE-The-FDA-Reminds-Patients-and-Health-Care-Providers-of-the-Importance-of-At-Least-YearlyOriginal Title: UPDATE: The FDA reminds patients and health care providers of the importance of at least yearly, lifelong follow-up with use of Endologix AFX endovascular AAA graft systems: FDA Safety Communication.
Reference: US Food and Drug Administration. Published and accessed on: December 4, 2020.
Subscribe to our weekly newsletter
Get the latest scientific articles on interventional cardiology





