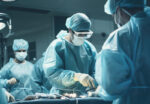
Treating tricuspid regurgitation (TR) has gained increasing importance in interventionism. There is abundant literature on the negative natural evolution of this condition and its unfavorable prognosis, as well as the clinical challenges associated to optimal medical treatment. Among its percutaneous treatment options, both edge-to-edge repair devices (TEER), as showed in TriValve and TRILUMINATE trials, and...
New Advances in Mitral Regurgitation Devices with Promising Results
Mitral regurgitation (MR) is a frequent valvulopathy and when the optimal medical treatment at maximum tolerated doses is not enough, surgery is the first course of action. And even though edge-to-edge repair with clips has seen substantial improvement, it is currently reserved for those at high risk of surgery. However, there is an increasing number...
MitraScore: What Does the Final Result of Edge-to-Edge Treatment of Mitral Regurgitation Provide?
Edge-to-edge treatment has proven to be a safe and effective technique in follow-up, decreasing mortality and hospitalizations for heart failure when a good result is achieved. The MitraScore was developed to assess the outcome at the end of the procedure. In that sense, results <3 represent mild mitral regurgitation, which would be related to lower...
Mitral Trial: 2-Year Followup
At present, an important group of patients with mitral valve disease are at high surgical risk, especially those with deteriorated bio-prosthesis, severe mitral annulus calcification or those who had received mitral annuloplasty. Percutaneous treatment with balloon expandable valves is a valid option to treat these patients, seeing as their 30 day and one-year outcomes look...
Valve-in-Valve Shows Good Evolution after 2 Years
The degeneration of bioprostheses in aortic position occurs at approximately after 10 to 15 years. In this scenario, the treatment of choice used to be redo surgery, but with the evolution of transcatheter aortic valve replacement (TAVR), it became a valid alternative with a level IIa B evidence. While there are currently multiple analyses of...
TCT 2022 | CLASP II D TRIAL: Transcatheter Edge-to-Edge Repair for Mitral Regurgitation in Patients at Prohibitive Risk
The CLASP IID study was a prospective randomized study to assess the efficacy and effectiveness of the PASCAL transcatheter valve repair system compared with the MitraClip device for patients with degenerative mitral regurgitation (MR) at prohibitive surgical risk. The study included Patients at prohibitive surgical risk, candidates for both devices, with MR 3+ to 4+,...
SOLACI-SOCIME 2022 – MITRAL VALVE Session
✔️ MITRAL VALVE Session ✔️ SOLACI-SOCIME 2022 Scientific Session 👨🏫 Content: 00:07 – Welcoming Words- Dr. Guering Eid Lidt (MEX) 01:26 – Mitral valve replacement – Dr. Ignacio Amat (ESP) 18:15 – Transcatheter treatment of mitral valve regurgitation – Dr. Luis Nombela Franco (ESP) 34:57 – Mitral valve-in-valve – Dr. Guering Eid Lidt (MEX)
The Best of the SOLACI-SOCIME 2022 Main Arena: Mitral Valve
During the “Mitral Valve” session (on the first day of the SOLACI-SOCIME 2022 Congress), Dr. Ignacio Amat Santos (SPA) delivered a great talk on transcatheter mitral valve replacement (TMVR). During his presentation, he discussed different scenarios for transcatheter replacement: mitral valve-in-vale, mitral valve-in-ring, mitral valve in MAC, and new devices for non-calcified native valves. When...
EuroPCR 2022 | Changes in Cardiac Damage after Surgical Aortic Valve Replacement
This study presented at EuroPCR 2022 analyzed the PARTNER study pool and included 1974 patients with a complete echocardiogram. Surgical risk was 17.3% extreme/inoperable, 54.3% intermediate and 28.4% low. 60% received transcatheter aortic valve replacement (TAVR) and the rest surgical aortic valve replacement (SAVR). 6.1% of patients were in stage 0 (no damage), 14.5% were...
New Valvular Heart Disease Guidelines with Key TAVI and Mitral Regurgitation Updates
Valvular heart disease management guidelines were updated last week by the AHA and the ACC. The last complete version had been published in 2014; therefore many of its recommendations have become obsolete before the latest evidence. The studies that made the FDA approve TAVI for low-risk patients are probably the most important incorporations. They also...